A drug that’s at the moment used for the therapy of psoriasis has been discovered to be efficient in treating the early phases of type-1 diabetes in kids and adolescents, finds a brand new scientific trial led by Cardiff College.
The brand new research has proven that Ustekinumab, a longtime immunotherapy that has been used to deal with psoriasis since 2009, is efficient in preserving the physique’s capability to supply insulin in type-1 diabetes—bringing the purpose of managing type-1 diabetes with out insulin a step nearer.
The research by Cardiff College, Kings School London, Swansea College and the College of Calgary, unlocked new insights into figuring out the precise immune cells (Th17 cells) that trigger type-1 diabetes and additional established a task for immunotherapies in curbing the destruction of insulin-producing cells.
The scientific trial examined the psoriasis therapy in 72 adolescents between the ages of 12 and 18 with recent-onset type-1 diabetes. The findings from the research have been revealed in Nature Drugs on July 30, 2024.
“Kind-1 diabetes happens when the physique’s immune system assaults and destroys the cells of the physique that produce insulin. This ultimately leaves the particular person depending on insulin injections. Researchers are actually creating methods to sluggish or halt the immune system assault. If such remedies could be began early, earlier than all of the insulin-making cells are misplaced, this might forestall or scale back the necessity for insulin,” says Dr. Danijela Tatovic.
Researchers hope that immunotherapy will present solutions for sufferers sooner or later, focusing on the physique’s immune system to decelerate the destruction of the cells that produce insulin. This treats the underlying immune course of moderately than correcting insulin ranges.
Ustekinumab is an injection therapy which sufferers can provide themselves at residence, used successfully within the therapy of greater than 100,000 sufferers with immune circumstances, together with extreme psoriasis, psoriatic arthritis, extreme Crohn’s illness and extreme ulcerative colitis.
This research demonstrated that Ustekinumab may protect very important insulin-producing cells. The researchers additionally recognized the precise immune cells that trigger this destruction, enabling exact and focused therapies to maximise advantages and decrease unwanted side effects.
Professor Tim Tree, King’s School London, stated, “We have now discovered that Ustekinumab reduces the extent of a tiny group of immune cells within the blood referred to as Th17.1 cells. These cells make up only one in 1,000 of blood immune cells, however they appear to play an necessary function in destroying insulin-producing cells.
“This explains why Ustekinumab has so few side-effects. It targets the trouble-making cells, whereas leaving 99% of the immune system intact—a terrific instance of precision drugs.”
“We examined this therapy in kids and adolescents who already wanted insulin therapy. It could be higher if we may deal with them at an earlier stage, whereas the youngsters are nonetheless effectively, and stop them needing insulin. Fortunately, Ustekinumab has a ok security document to be thought-about to be used in kids at this early stage,” says Professor Colin Dayan.
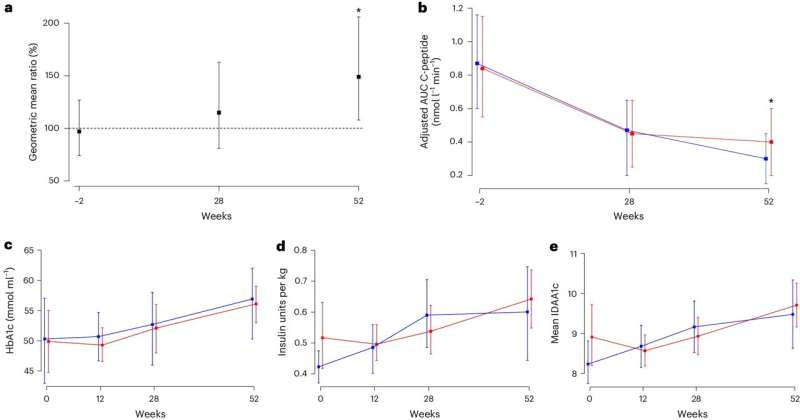
The usage of Ustekinumab was proven to lower the harmful affect of Th17 immune cells on cells that produce insulin. After 12 months of utilizing Ustekinumab, the researchers discovered that C-peptide ranges—an indication that the physique is producing insulin—had been 49% increased. This scientific trial additionally offers the primary scientific trial-based proof for the function of Th17 cells in type-1 diabetes.
Whereas the trial demonstrates the good thing about utilizing Ustekinumab to deal with type-1 diabetes, additional scientific trials are required to substantiate this discovering and to work out which sufferers would profit most from the therapy.
“It’s now potential with a easy finger-prick antibody check to detect kids who will develop type-1 diabetes years earlier than they want insulin. Combining screening on this approach with early therapy with Ustekinumab appears a really promising strategy to stopping the necessity for insulin. Additional trials can be wanted to substantiate this,” says Peter Taylor.
Extra info:
Danijela Tatovic et al, Ustekinumab for sort 1 diabetes in adolescents: a multicenter, double-blind, randomized part 2 trial, Nature Drugs (2024). DOI: 10.1038/s41591-024-03115-2
Cardiff College
Quotation:
Psoriasis drug reveals promise for treating childhood diabetes (2024, July 30)
retrieved 30 July 2024
from
This doc is topic to copyright. Aside from any truthful dealing for the aim of personal research or analysis, no
half could also be reproduced with out the written permission. The content material is offered for info functions solely.







